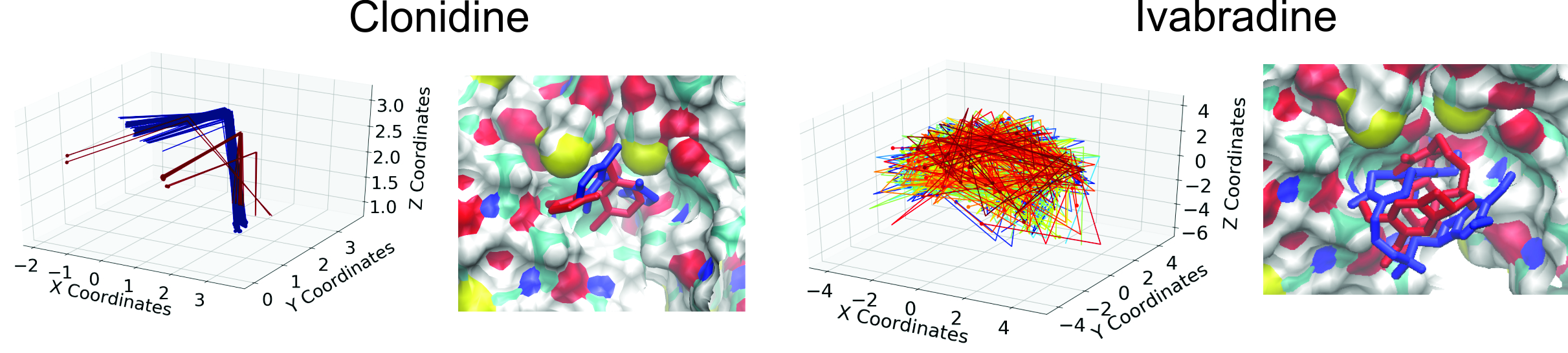
Characterization of drug binding within the HCN1 channel pore.
(Tanguay, J., Callahan, K.M., and D'Avanzo, N., Scientific Reports 2019)
Block of HCN channels slows the heart rate and is currently used to treat angina. However, HCN block also provides a promising approach to the treatment of neuronal disorders including epilepsy and neuropathic pain. While several molecules that block HCN channels have been identified, including clonidine and its derivative alinidine, lidocaine, mepivacaine, bupivacaine, ZD7288, ivabradine, zatebradine, and cilobradine,
their low affinity and lack of specificity prevents wide-spread use. We used computational docking experiments to assess the binding sites and mode of binding of these inhibitors against the recently solved atomic structure of human HCN1 channels, and a homology model of the open pore derived from a closely related CNG channel. We identify a possible hydrophobic groove in the pore cavity that plays an important role in conformationally restricting the location and orientation of drugs bound to the inner vestibule.
Our results also help explain the molecular basis of the low-affinity binding of these inhibitors, paving the way for the development of higher affinity molecules.
Read More...
Read More ...
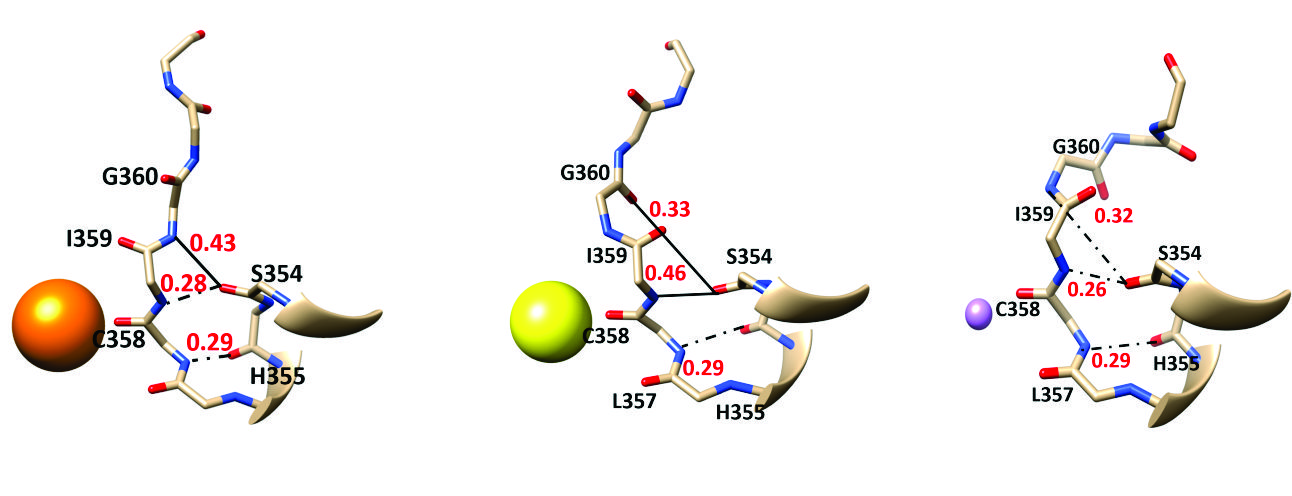
Ion behavior in the selectivity filter of HCN1 channels (Ahrari S, Ozturk TN, D'Avanzo N., Biophys J 2022)
Hyperpolarization-activated cyclic-nucleotide gated channels (HCNs) are responsible for the generation of pacemaker currents (If or Ih) in cardiac and neuronal cells. Despite the overall structural similarity to voltage-gated potassium (Kv) channels, HCNs show much lower selectivity for K+ over Na+ ions.
This increased permeability to Na+ is critical to their role in membrane depolarization. HCNs can also select between Na+ and Li+ ions. Here, we investigate the unique ion selectivity properties of HCNs using molecular-dynamics simulations. Our simulations suggest that the HCN1 pore is flexible and dilated compared with Kv channels with only one stable ion binding site within the selectivity filter. We also observe that ion coordination and hydration differ within the HCN1 selectivity filter compared with those in Kv and cyclic-nucleotide gated channels. Additionally, the C358T mutation further stabilizes the symmetry of the binding site and provides a more fit space for ion coordination, particularly for Li+.
Read More...
Read More ...
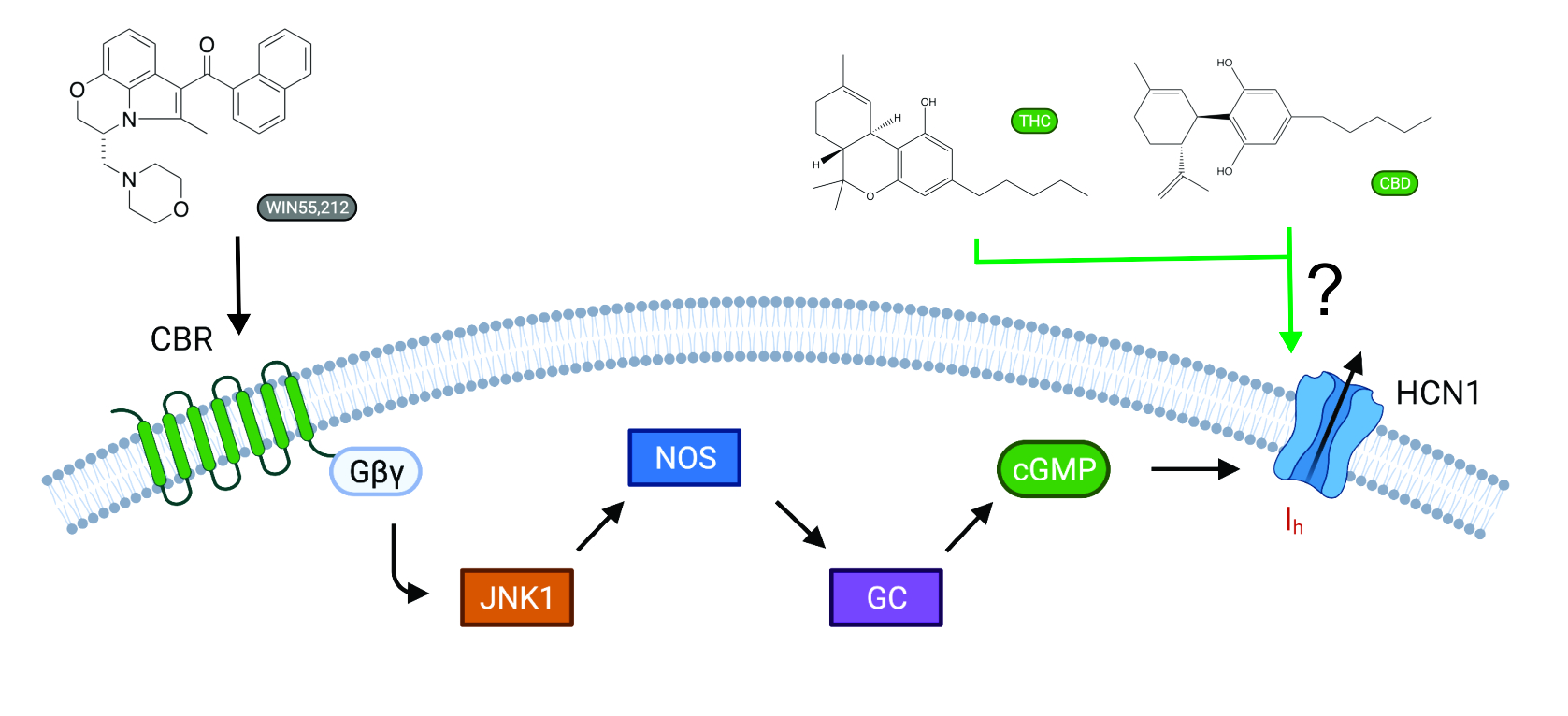
Direct Regulation of Hyperpolarization-Activated Cyclic-Nucleotide Gated (HCN1) Channels by Cannabinoids. (Mayar S, et al., Front Mol Neurosci 2022)
Cannabinoids are a broad class of molecules that act primarily on neurons, affecting pain sensation, appetite, mood, learning, and memory. In addition to interacting with specific cannabinoid receptors (CBRs), cannabinoids can directly modulate the function of various ion channels. Here, we examine whether cannabidiol (CBD) and Δ9-tetrahydrocannabinol (THC), the most prevalent phytocannabinoid
s in Cannabis sativa, can regulate the function of hyperpolarization-activated cyclic-nucleotide-gated (HCN1) channels independently of CBRs. HCN1 channels were expressed in Xenopus oocytes since they do not express CBRs, and the effects of cannabinoid treatment on HCN1 currents were examined by a two-electrode voltage clamp. We observe opposing effects of CBD and THC on HCN1 current,
CBD acting to stimulate HCN1 function, while THC inhibited current. These effects persist in HCN1 channels lacking the cyclic-nucleotide binding domain (HCN1ΔCNBD). However, changes to membrane fluidity, examined by treating cells with TX-100, inhibited HCN1 current had more pronounced effects on the voltage-dependence and kinetics of activation than THC,
suggesting this is not the primary mechanism of HCN1 regulation by cannabinoids... Read more...
Read More
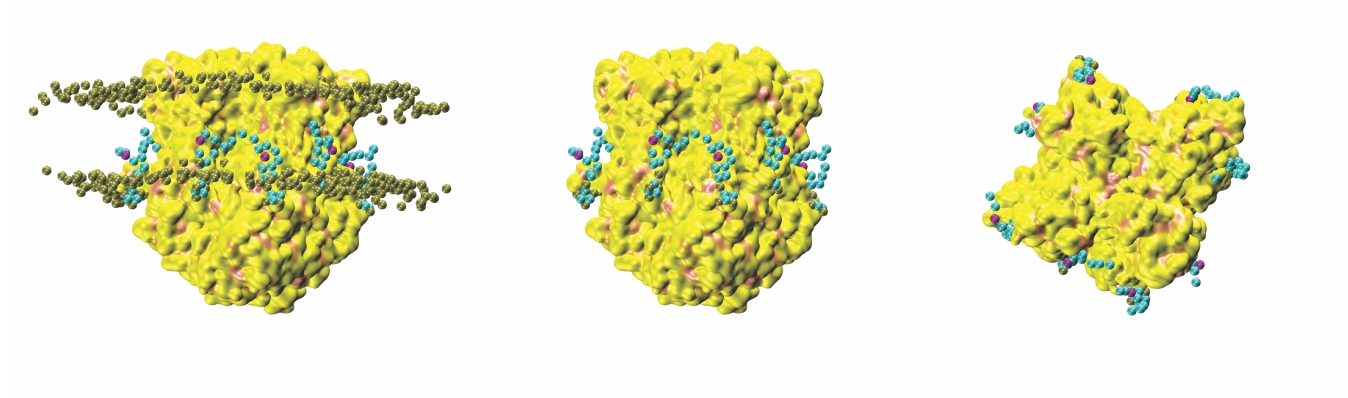
Computational Prediction of Phosphoinositide Binding to HCN Channels (Claveras Cabezudo A, Feriel Khoualdi A, D'Avanzo N., Front Physiol 2022)
Protein-lipid interactions are key regulators of ion channel function. Numerous ion channels, including hyperpolarization-activated cyclic-nucleotide gated (HCN) channels have been shown to be regulated by phosphoinositides (PIPs), with important implications in cardiac and neuronal function. Specifically, PIPs have been shown to enhance HCN activation. Using computational approaches, we aim to
identify potential binding sites for HCN1-PIP interactions. Computational docking and coarse-grained simulations indicate that PIP binding to HCN1 channels is not well coordinated, but rather occurs over a broad surface of charged residues primarily in the HCN-domain, S2 and S3 helices that can be loosely organized in 2 or 3 overlapping clusters. Thus, PIP-HCN1 interactions are more resembling of
electrostatic interactions that occur in myristoylated alanine-rich C kinase substrate (MARCKS) proteins, than the specifically coordinated interactions that occur in pleckstrin homology domains (PH domains) or ion channels such as inward rectifier potassium (Kir) channels...Read more...
Read More